Patient Focused.
- Home
- Wound Care Professionals
- Clinical
Advancing science to improve outcomes is what drives us. We are relentless in our pursuit of better solutions, fueled by a passion to move the science of treating wounds forward. Our growing body of published research continues to tell a powerful story of success—one that leads to restored health and renewed lives.
Aseptic Processing
Aseptic Processing
Placental Matrices
Dehydrated human amnion/chorion-derived allograft (dHACA)



Publications
Placental Publications


DiDomenico, Lawrence A. DPM, FACFAS; Orgill, Dennis P. MD, PhD; Galiano, Robert D. MD; Serena, Thomas E. MD, FACS; Carter, Marissa J. PhD, MA; Kaufman, Jarrod P. MD, FACS; Young, Nathan J. DPM, Zelen, Charles M. DPM, FACFAS, FACFAOM, Aseptically Processed Placental Membrane Improves Healing of Diabetic Foot Ulcerations: Prospective, Randomized Clinical Trial. Plast Reconstr Surg Glob Open, 2016 Oct, 12:4(10):e1095, DOI: 10.1097/GOX.0000000000001095

Case Studies
MTF Biologics Wound Care tissue forms have been designed to address a wide variety of wounds.
Please select any of the case studies presented below to learn more about our successes in application.
Placental Case Studies
DiDomenico, Lawrence A. DPM, FACFAS; Kaufman, Jarrod P. MD, FACS; Serena, Thomas E. MD, FACS, Use of AmnioBand™ Membrane, a Human Amniotic Membrane Allograft, in the Management of Chronic Non-Healing Diabetic Foot Ulcers
Scientific/Clinical Posters
We have presented our findings at conferences and meetings across the country. Click on any of the items below to view and download scientific studies of our tissue forms in easy-to-reference posters.
Placental Scientific Posters
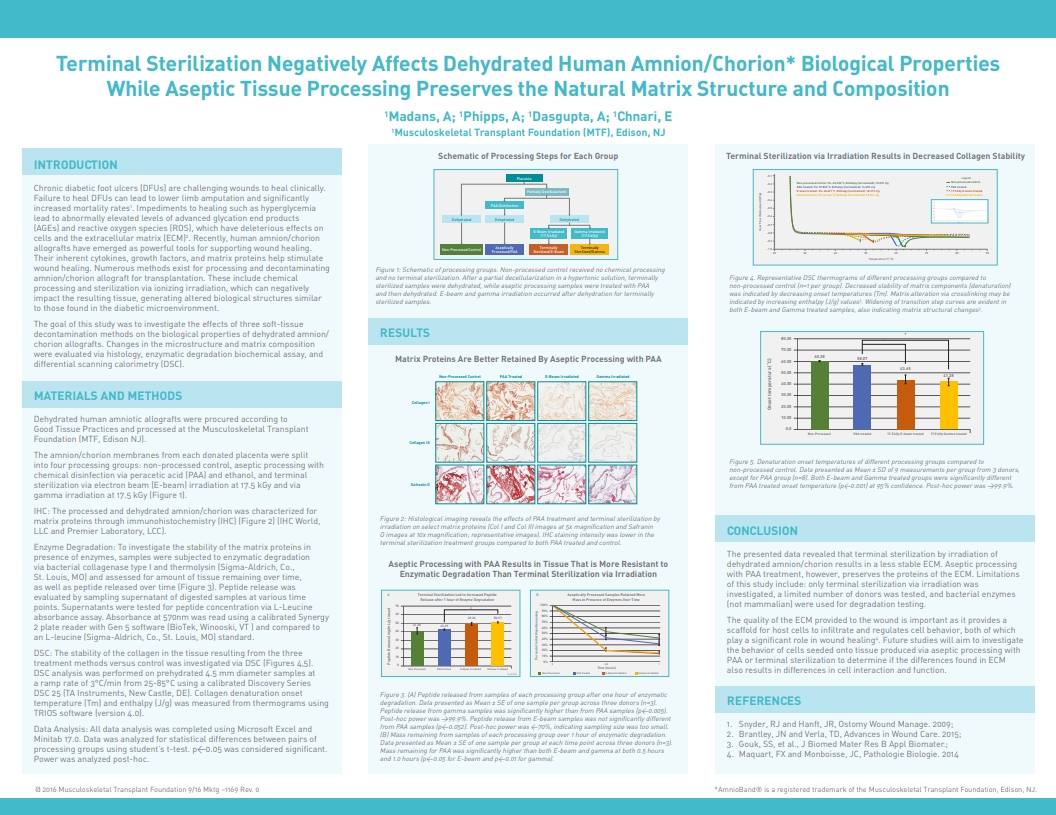
Madans, A; Phipps, A; Dasgupta, A.; Chnari, E., Terminal sterilization negatively affects dehydrated human amnion/chorion biological properties while aseptic tissue processing preserves the natural matrix structure and composition, 2016 Fall
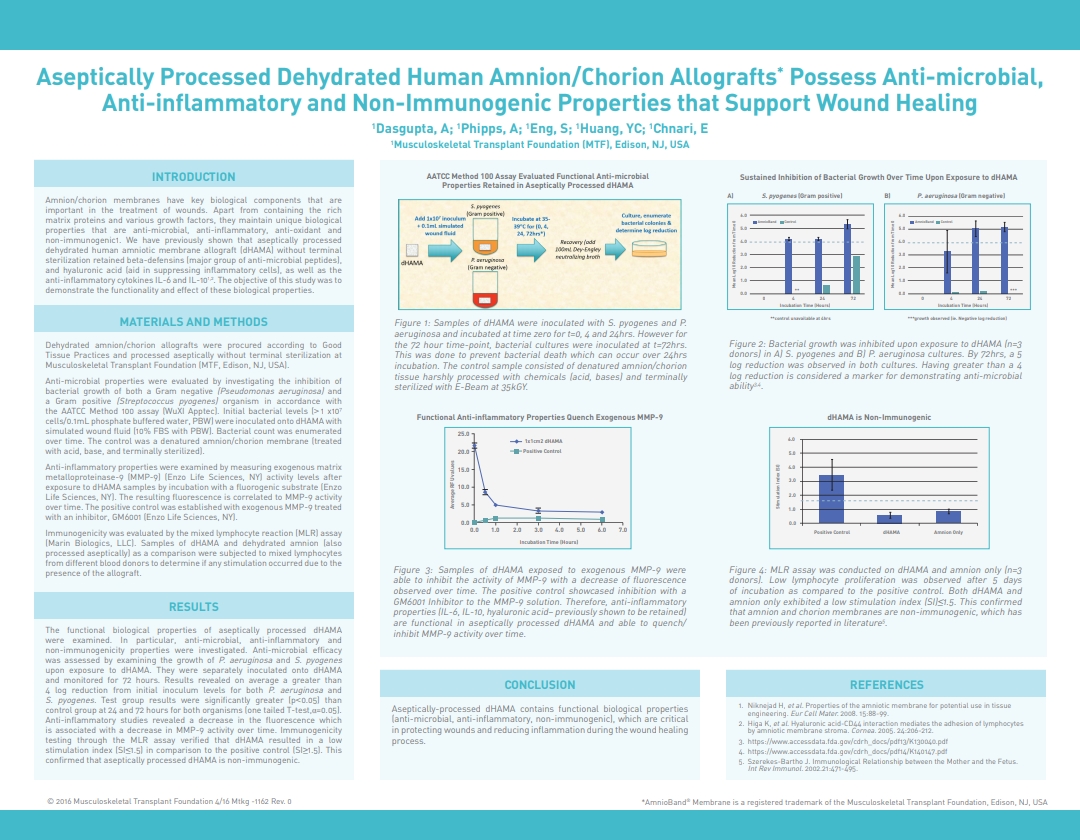
Dasgupta, A.; Phipps, A.; Eng, S.; Huang, Y.C.; Chnari, E., Aseptically Processed Dehydrated Human Amnion/Chorion Allografts Possess Anti-microbial, Anti-inflammatory and Non-immunogenic Properties that Support Wound Healing, 2016 Spring
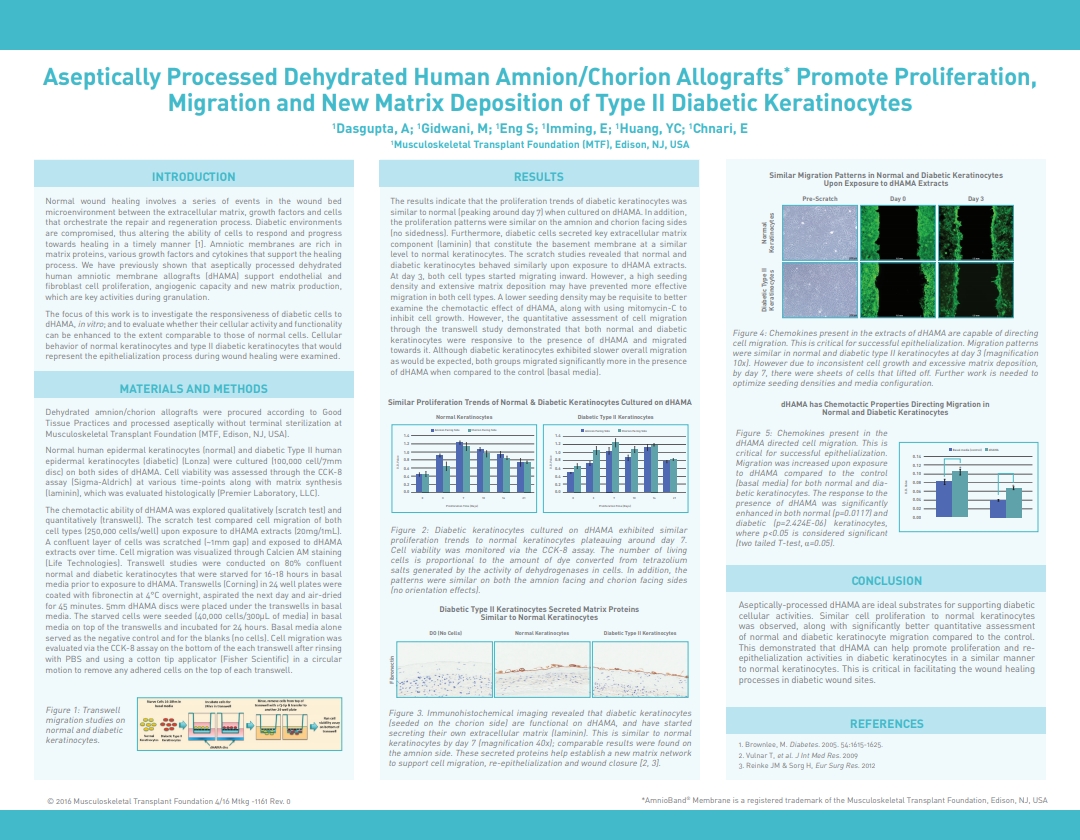
Dasgupta, A.; Huang, Y.C.; Gidwani, M.; Imming, E.; Eng, S; Chnari, E., Aseptically Processed Dehydrated Human Amnion/Chorion Allografts Promote Proliferation, Migration and New Matrix Deposition of Type II Diabetic Keratinocytes, 2016 Spring
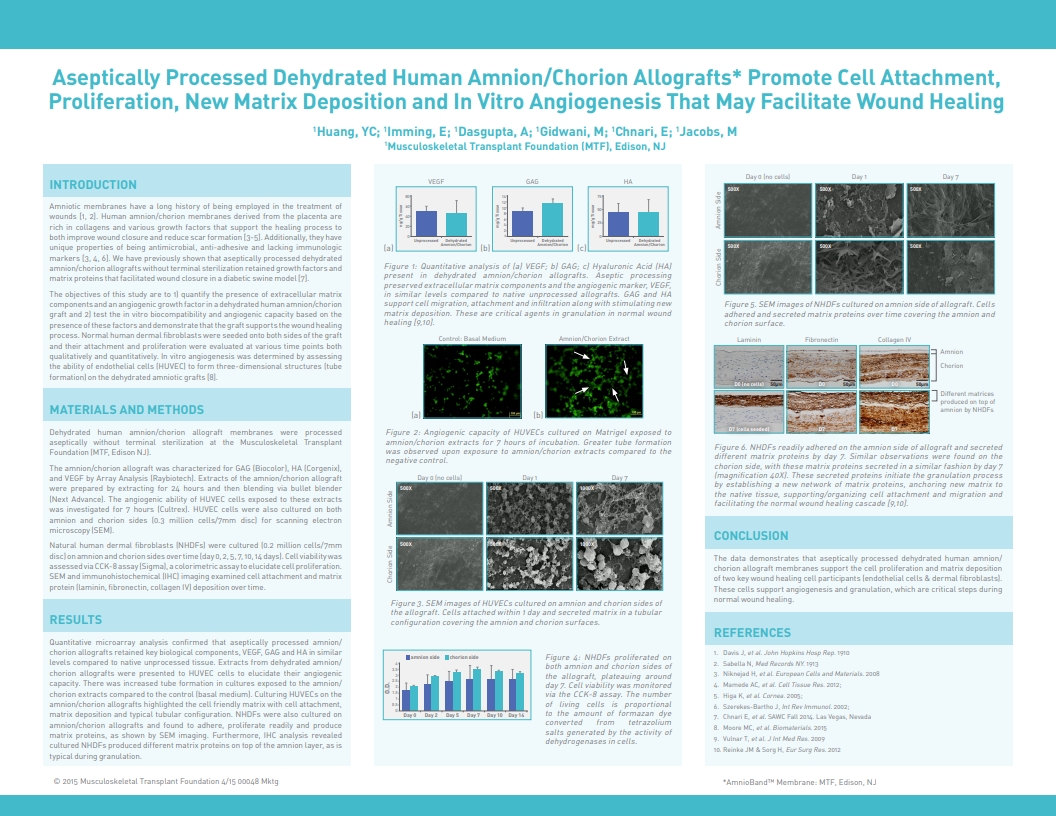
Huang, Y.C.; Imming, E.; Dasgupta, A.; Gidwani, M.; Chnari, E.; Jacobs, Allen M. DPM, Aseptically Processed Dehydrated Human Amnion/Chorion Allografts Promote Cell Attachment, Proliferation, New Matrix Deposition and In Vitro Angiogenesis That Supports Wound Healing, 2016 Spring
Chnari, E.; Dasgupta, A.; Huang, Y.C.; Jacobs, Allen M. DPM, Aseptically Processed Dehydrated Human Amnion/Chorion Allograft Promotes Healing of Chronic Wounds in a Diabetic Swine Model, 2014 Fall
Dermal Matrices
Human reticular acellular dermal matrix (HR-ADM)



Publications
Dermal Publications

Zelen, Charles M. DPM, FACFAS, FACFAOM; Orgill, Dennis P. MD, PhD; Serena, Thomas E. MD, FACS; Galiano, Robert D. MD; Carter, Marissa J. PhD, MA; DiDomenico, Lawrence A. DPM, FACFAS; Kaufman, Jarrod P. MD, FACS; Keller, K.; Li, W.W., An aseptically processed, acellular, reticular, allogenic human dermis improves healing in diabetic foot ulcers: A prospective, randomised, controlled, multicentre follow‐up trial, Int Wound J., 2018 Apr, DOI: 10.1111/iwj.12920

Dasgupta, A.; Orgill, Dennis P. MD, PhD; Galiano, Robert D. MD; Zelen, Charles M. DPM, FACFAS, FACFAOM; Huang, Y.C.; Chnari, E.; Li, W.W., A Novel Reticular Dermal Graft Leverages Architectural and Biological Properties to Support Wound Repair, Plast Reconstr Surg Glob Open, 2016 Oct, 4(10):p e1065, DOI: 10.1097/GOX.0000000000001065
Case Studies
MTF Biologics Wound Care tissue forms have been designed to address a wide variety of wounds.
Please select any of the case studies presented below to learn more about our successes in
Dermal Case Studies
Zelen, Charles M. DPM, FACFAS, FACFAOM; Kaufman, Jarrod P. MD, FACS; Lang, A. DPM, Use of AlloPatch® Pliable, a Human Acellular Dermal Matrix, as an Adjunctive Therapy for Chronic Non-Healing Diabetic Foot Ulcer
Scientific/Clinical Posters
We have presented our findings at conferences and meetings across the country. Click on any of the items below to view and download scientific studies of our tissue forms in easy-to-reference posters.
Dermal Scientific Posters
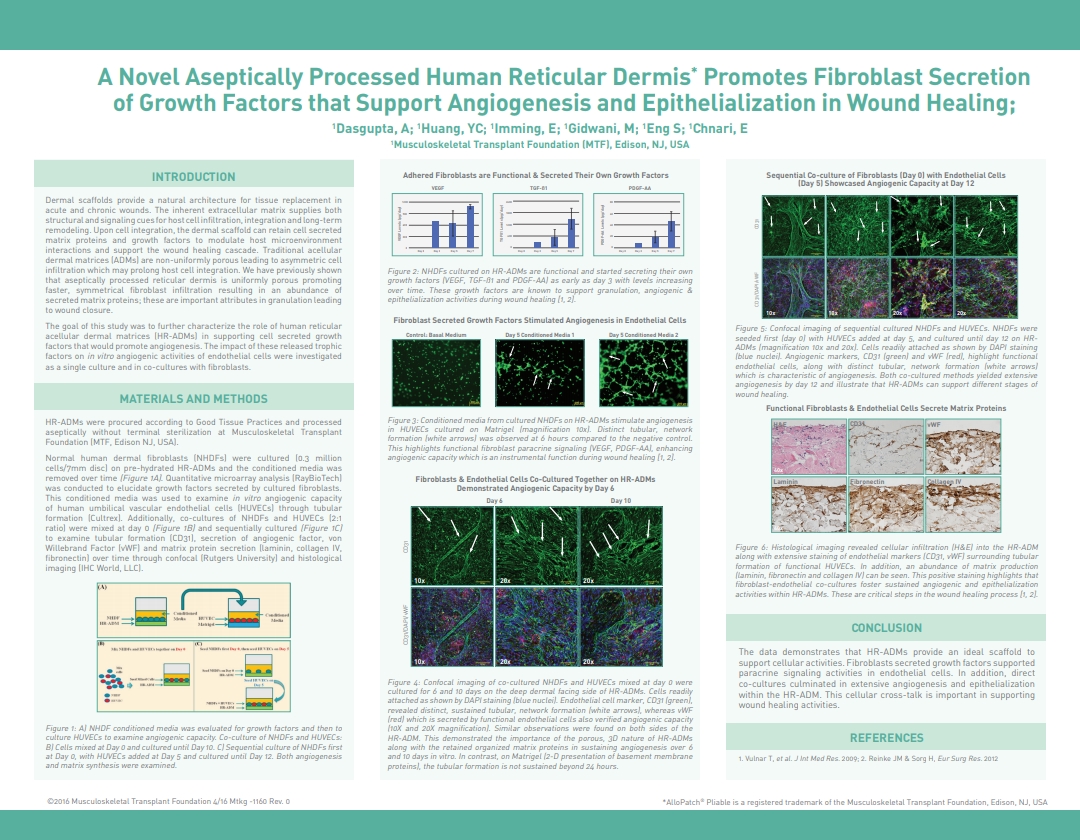
Dasgupta, A.; Huang, Y.C.; Imming, E.; Gidwani, M.; Eng, S.; Chnari, E., A Novel Aseptically Processed Human Reticular Dermis Promotes Fibroblast Secretion of Growth Factors that Support Angiogenesis and Epithelialization in Wound Healing, 2016 Spring
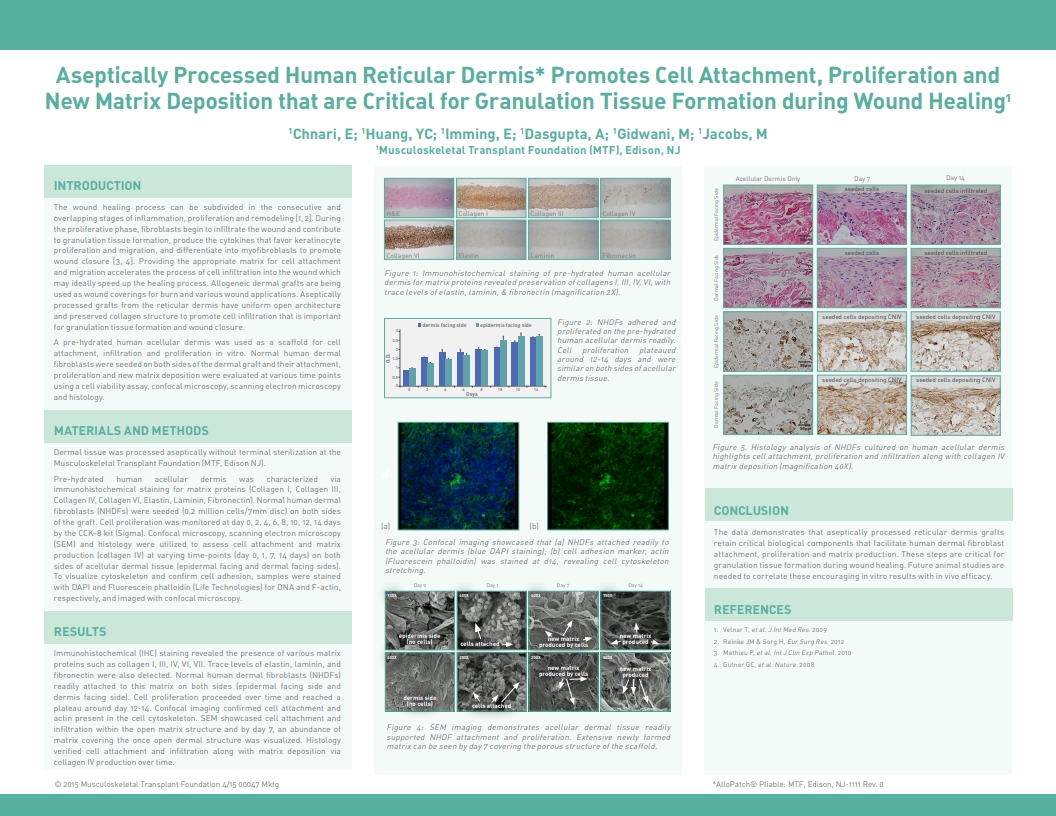
Chnari, E.; Huang, Y.C.; Imming, E.; Jacobs, Allen M. DPM, A Novel Design of Aseptically Processed Human Reticular Dermis Promotes Enhanced Cell Attachment, Proliferation and New Matrix Deposition in Wound Healing, 2015 Spring
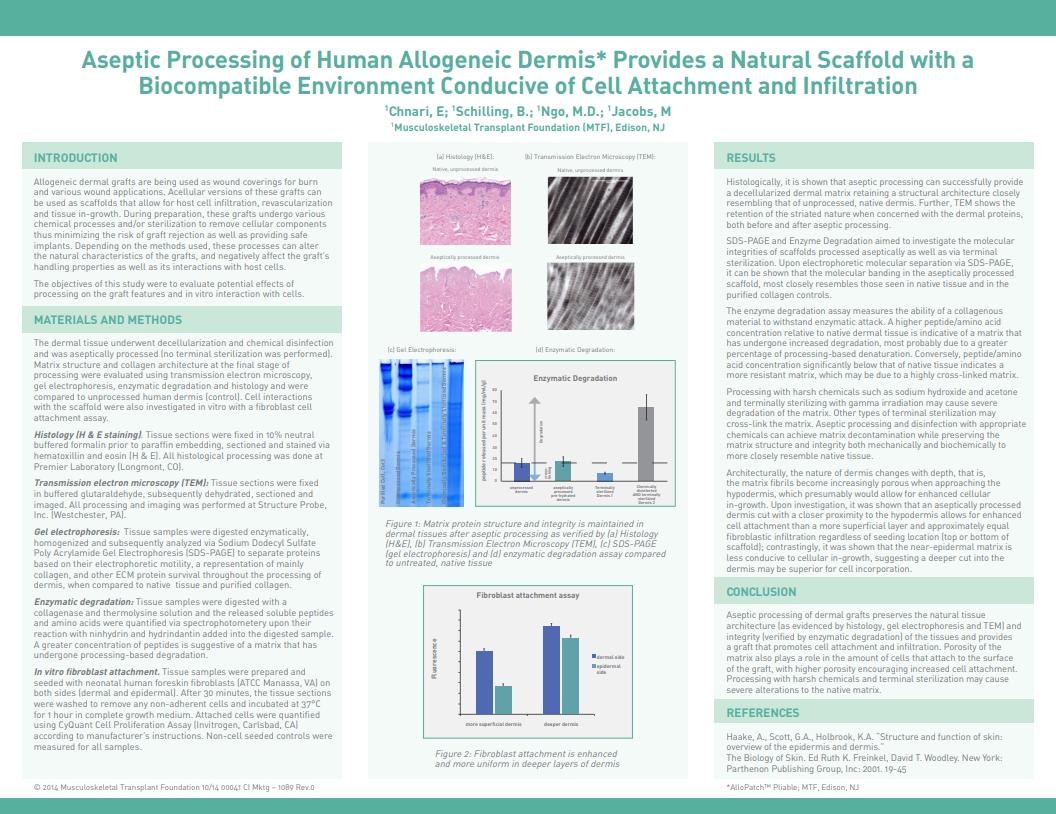
Chnari, E.; Shilling, B.; Ngo, M-D.; Jacobs, Allen M. DPM, Aseptic Processing of Human Allogeneic Dermis Provides a Natural Scaffold with a Biocompatible Environment Conducive of Cell Attachment and Infiltration, 2014 Fall
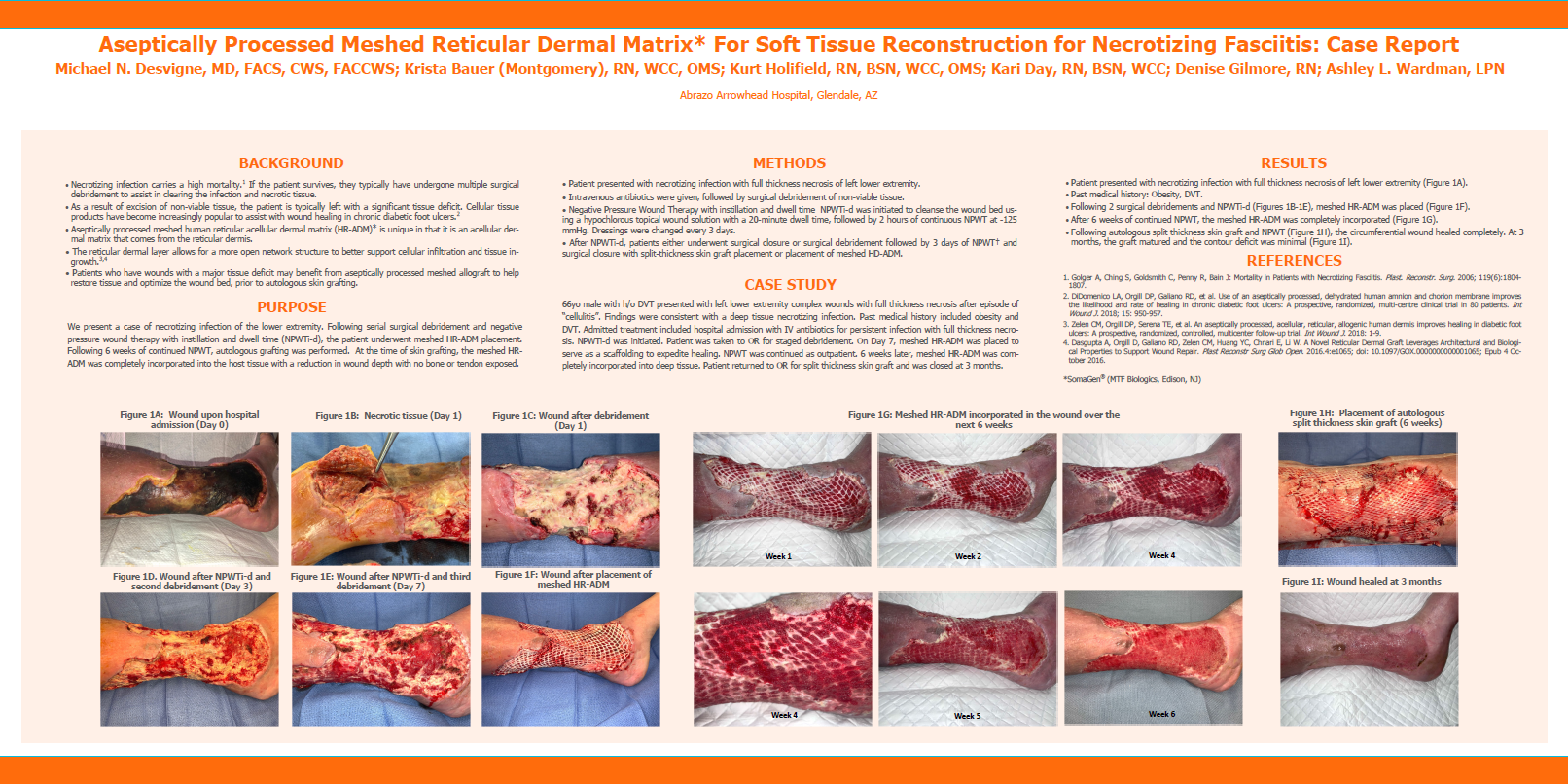
Desvigne, M.; Bauer, K.; Holifield, K.; Day, K.; Gilmore, D.; Wardman A., Aseptically Processed Meshed Reticular Dermal Matrix* For Soft Tissue Reconstruction for Necrotizing Fasciitis: Case Report, 2020 Fall
The clinical studies featured in this section are for informational purposes only.
For more information about clinical uses of Flex HD, please email MSL@mtf.org.
Please acknowledge receipt of this notice below.